47.If the molar concentration of PBI2 is .0015 mol/litre the concentration of iodide ions in g ion/litre is
![SOLVED: What volume (in mL) of a 2.40 mol L −1 magnesium bromide solution would contain 3.40 x 10^23 bromide [Br− (aq)] ions? A) 118 B) 12.3 C) 543 D) 58.9 E) 74.6 SOLVED: What volume (in mL) of a 2.40 mol L −1 magnesium bromide solution would contain 3.40 x 10^23 bromide [Br− (aq)] ions? A) 118 B) 12.3 C) 543 D) 58.9 E) 74.6](https://cdn.numerade.com/ask_previews/1a8be9cb-b10d-490e-9333-ae6f4d305d94_large.jpg)
SOLVED: What volume (in mL) of a 2.40 mol L −1 magnesium bromide solution would contain 3.40 x 10^23 bromide [Br− (aq)] ions? A) 118 B) 12.3 C) 543 D) 58.9 E) 74.6
What is H+in mol/L of a solution that is 0.20 M CH3COONa and 0.10 M in CH3COOH? (Ka for CH3COOH =1.8x10 5)

PPT - MOLARITY A measurement of the concentration of a solution PowerPoint Presentation - ID:1459950
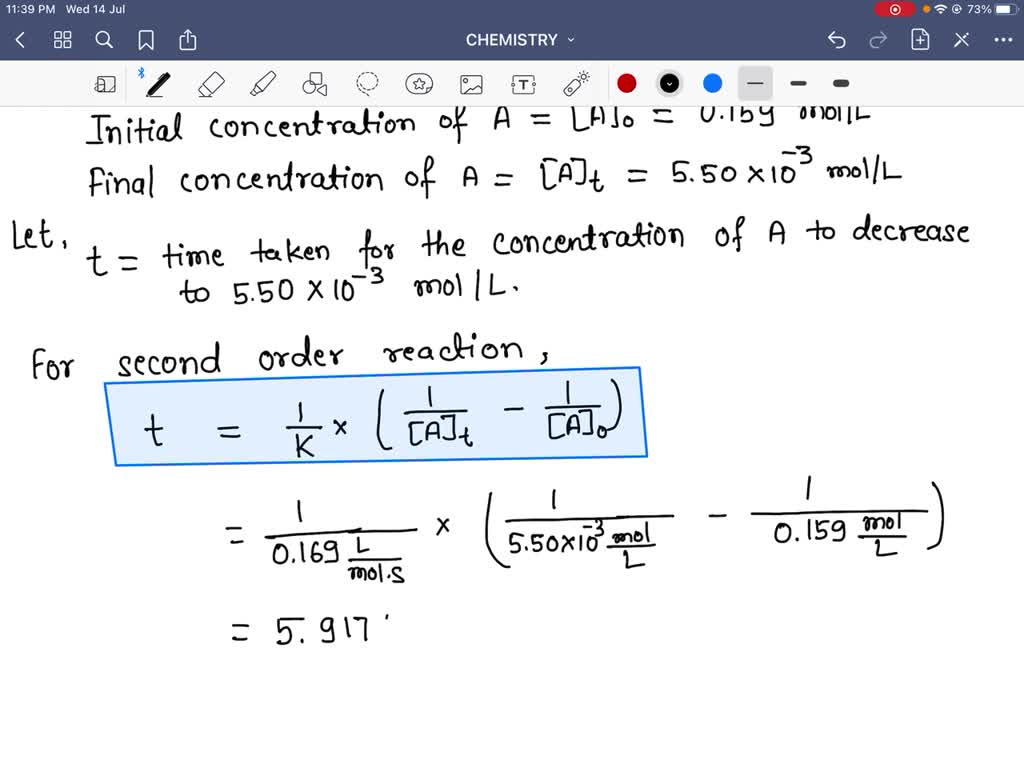
SOLVED:A reaction of the form a A ⟶Products is second order with a rate constant of 0.169 L /(mol ·s) . If the initial concentration of A is 0.159 mol / L,

What does Mol/l mean? - Definition of Mol/l - Mol/l stands for Mole Per Liter. By AcronymsAndSlang.com

Molarity 2. Molarity (M) this is the most common expression of concentration M = molarity = moles of solute = mol liters of solution L Units are. - ppt download











