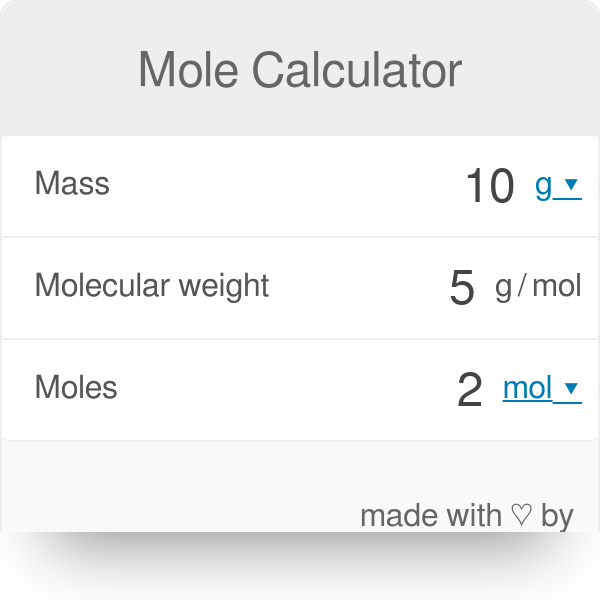
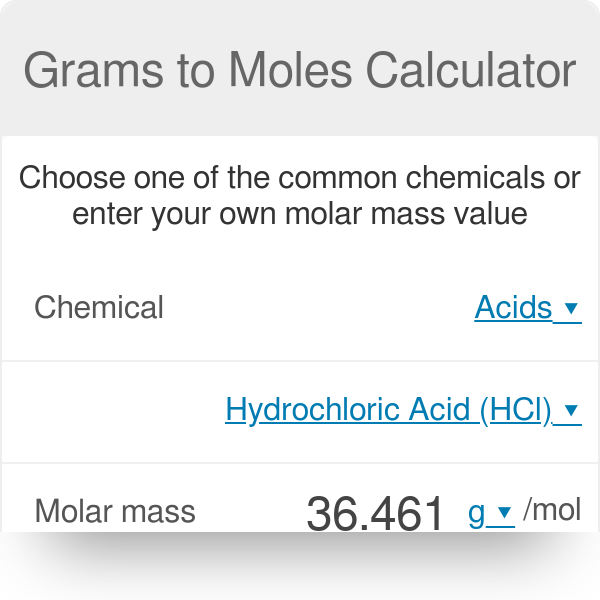
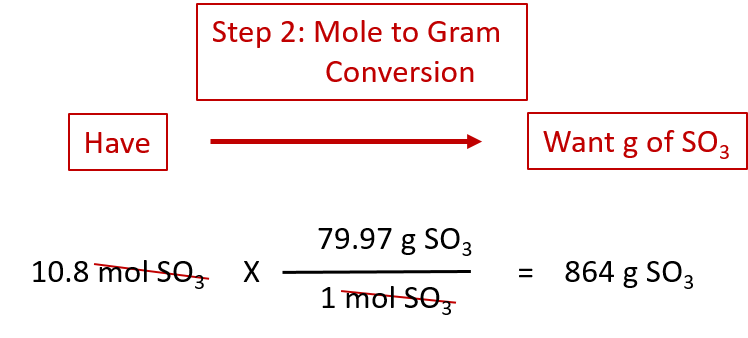
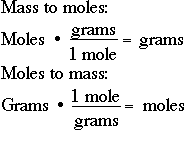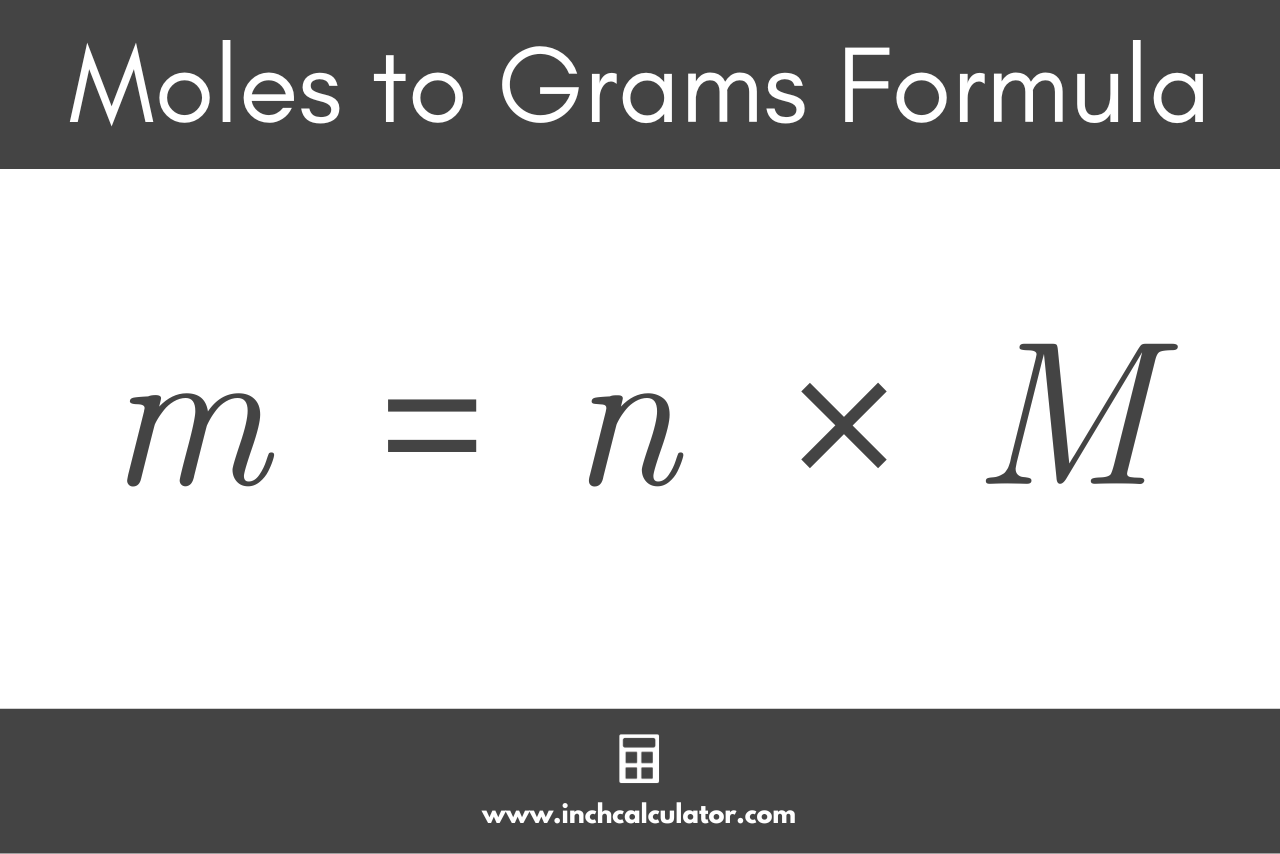enthalpy - Why is it sometimes kJ only, and in other times kJ/mol? What's the difference? - Chemistry Stack Exchange
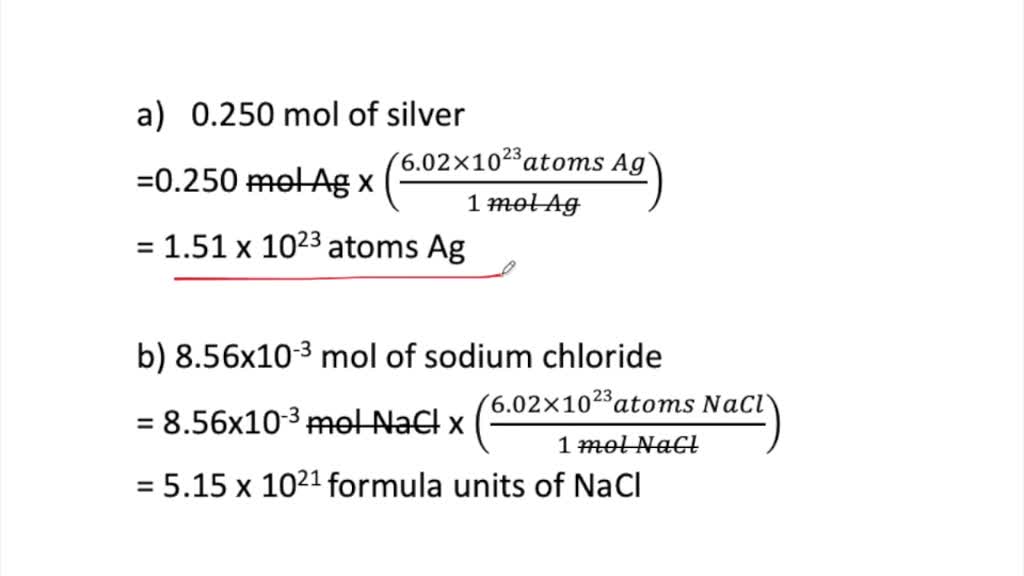
SOLVED:Determine the number of representative particles in each substance a. 0.250 mol of silver b. 8.56 ×10^-3 mol of sodium chloride c. 35.3 mol of carbon dioxide d. 0.425 mol of nitrogen (N2)