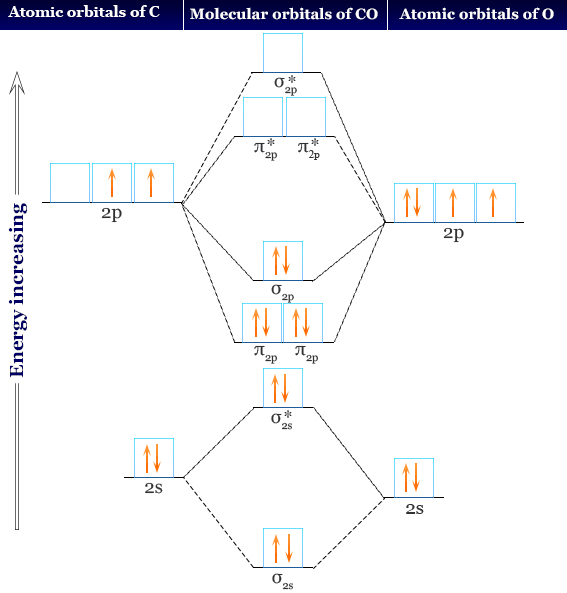
Temperature effect (10-55 • C) on the phenolphthalein (PHP);-CD-PHP... | Download Scientific Diagram

SOLVED: How many of each the following contained in 100 grams of CO2 (m=44.01)? calculate mol of C,O and O2

SOLVED: What is the freezing point, in °C, of a solution made with 1.07 mol of CHCl₃ in 530.0 g of CCl₄ (Kf = 29.8 °C/m, normal freezing point, Tf = -22.9 °C)?
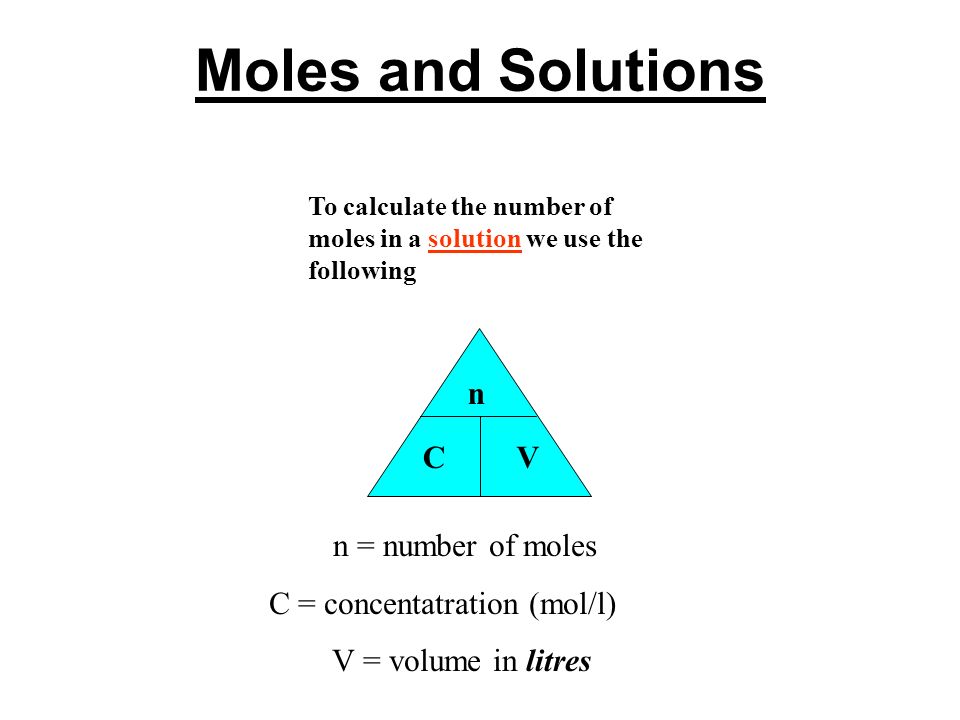
Moles and Solutions g n gfm To calculate the number of moles in a solution we use the following n CV n = number of moles C = concentatration (mol/l) V. - ppt download

Hydrogenation of fatty acids. a Oleic acid (0.5 mmol), SiNA-Pd (500 mol... | Download Scientific Diagram
1) At 2257 K and 1.00 atm total pressure, water is 1.77 per cent dissociated at equilibrium by way of the reaction 2 H2O(g) <
42. 1 mole of 'A', 1.5 mole of 'B' and 2 mole of 'C' are taken in a vessel of volume one litre . At equilibrium concentration of C is 0.5 mole/L.

The Mole And Concentration Formula Triangle Isolated On White Relationship Between Concentration Moles And Volume Cnv Stock Illustration - Download Image Now - iStock
![SOLVED: Conversion Factors 1 gal = 231 in' (exact) 1 atm 760 torr (exact) cm = ] mL (exact) 1 b = 453.59237 g (exact) 2.54 cm = L in (exact) 1 SOLVED: Conversion Factors 1 gal = 231 in' (exact) 1 atm 760 torr (exact) cm = ] mL (exact) 1 b = 453.59237 g (exact) 2.54 cm = L in (exact) 1](https://cdn.numerade.com/ask_images/98c41ed6b3e54a558ac16356f85004bd.jpg)