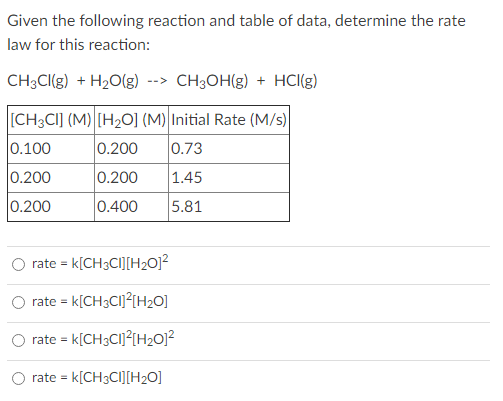
![SOLVED: Consider the reaction: CO(g)+H2O(g)⇌CO2(g)+H2(g) Kc=102 at 500 K A reaction mixture initially contains 0.130 M COand 0.130 M H2O. Part A What will be the equilibrium concentration of [CO]? [CO] = . SOLVED: Consider the reaction: CO(g)+H2O(g)⇌CO2(g)+H2(g) Kc=102 at 500 K A reaction mixture initially contains 0.130 M COand 0.130 M H2O. Part A What will be the equilibrium concentration of [CO]? [CO] = .](https://cdn.numerade.com/ask_previews/36628956-c06f-439b-80ff-a0f936ba3b67_large.jpg)
SOLVED: Consider the reaction: CO(g)+H2O(g)⇌CO2(g)+H2(g) Kc=102 at 500 K A reaction mixture initially contains 0.130 M COand 0.130 M H2O. Part A What will be the equilibrium concentration of [CO]? [CO] = .

SOLVED: A mixture of 0.466 M H2O , 0.481 M Cl2O , and 0.718 M HClO are enclosed in a vessel at 25°C . H2O(g)+Cl2O(g)↽−−⇀2HOCl(g)𝐾c=0.0900 at 25°C Calculate the equilibrium concentrations of
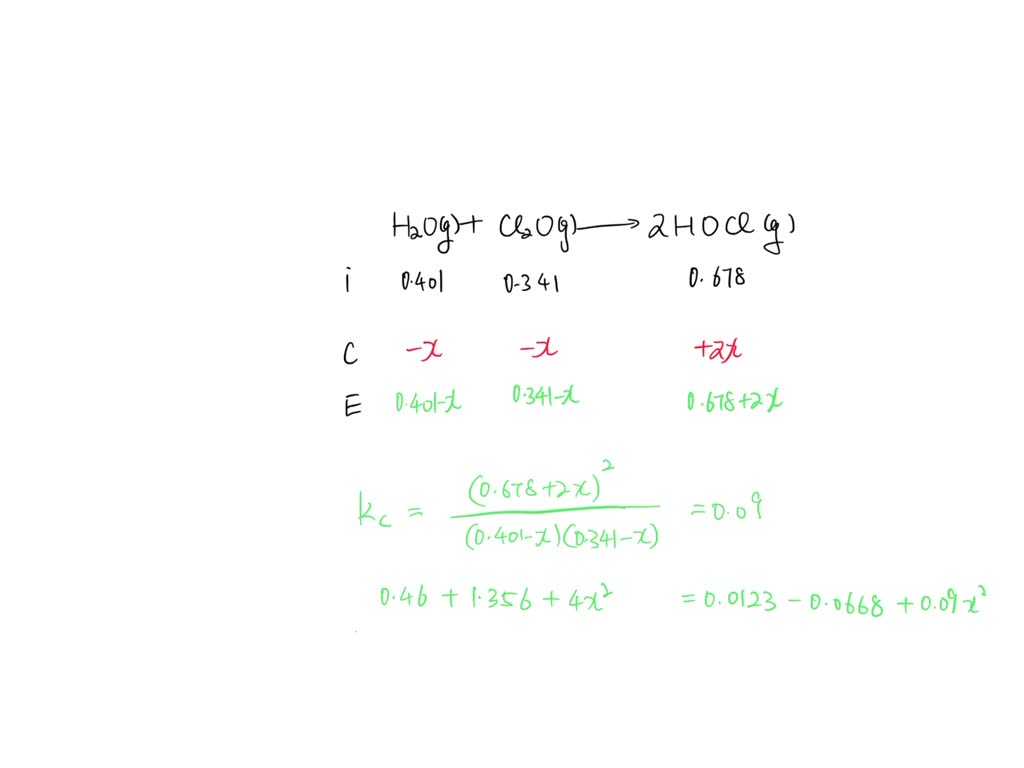
SOLVED: A mixture of 0.401 M H2O, 0.341 M Cl2O , and 0.678 M HClO are enclosed in a vessel at 25°C . H2O(g)+Cl2O(g)−⇀↽−2HOCl(g) Kc=0.0900 at 25°C Calculate the equilibrium concentrations of
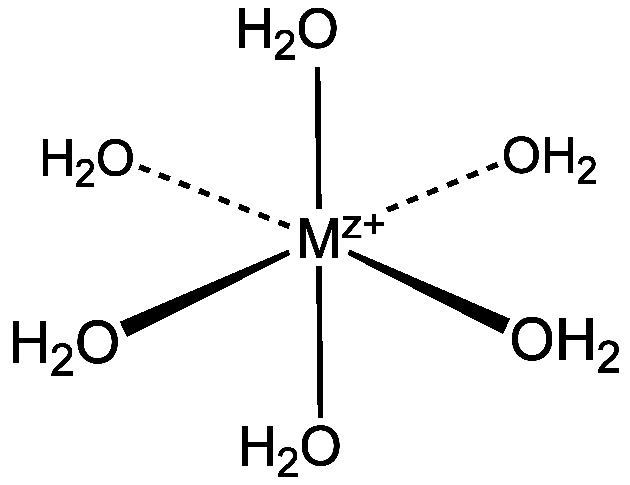
![Electronic Structure of 3d [M(H2O)6]3+ Ions from ScIII to FeIII: A Quantum Mechanical Study Based on DFT Computations and Natural Bond Orbital Analyses | Inorganic Chemistry Electronic Structure of 3d [M(H2O)6]3+ Ions from ScIII to FeIII: A Quantum Mechanical Study Based on DFT Computations and Natural Bond Orbital Analyses | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/ic001258t/asset/images/medium/ic001258tf00003.gif)