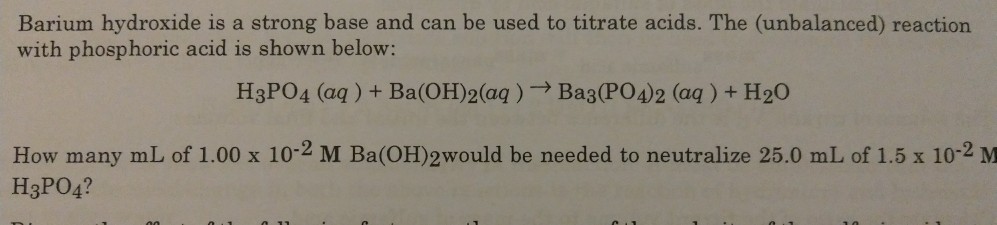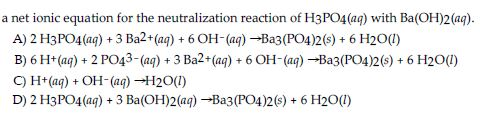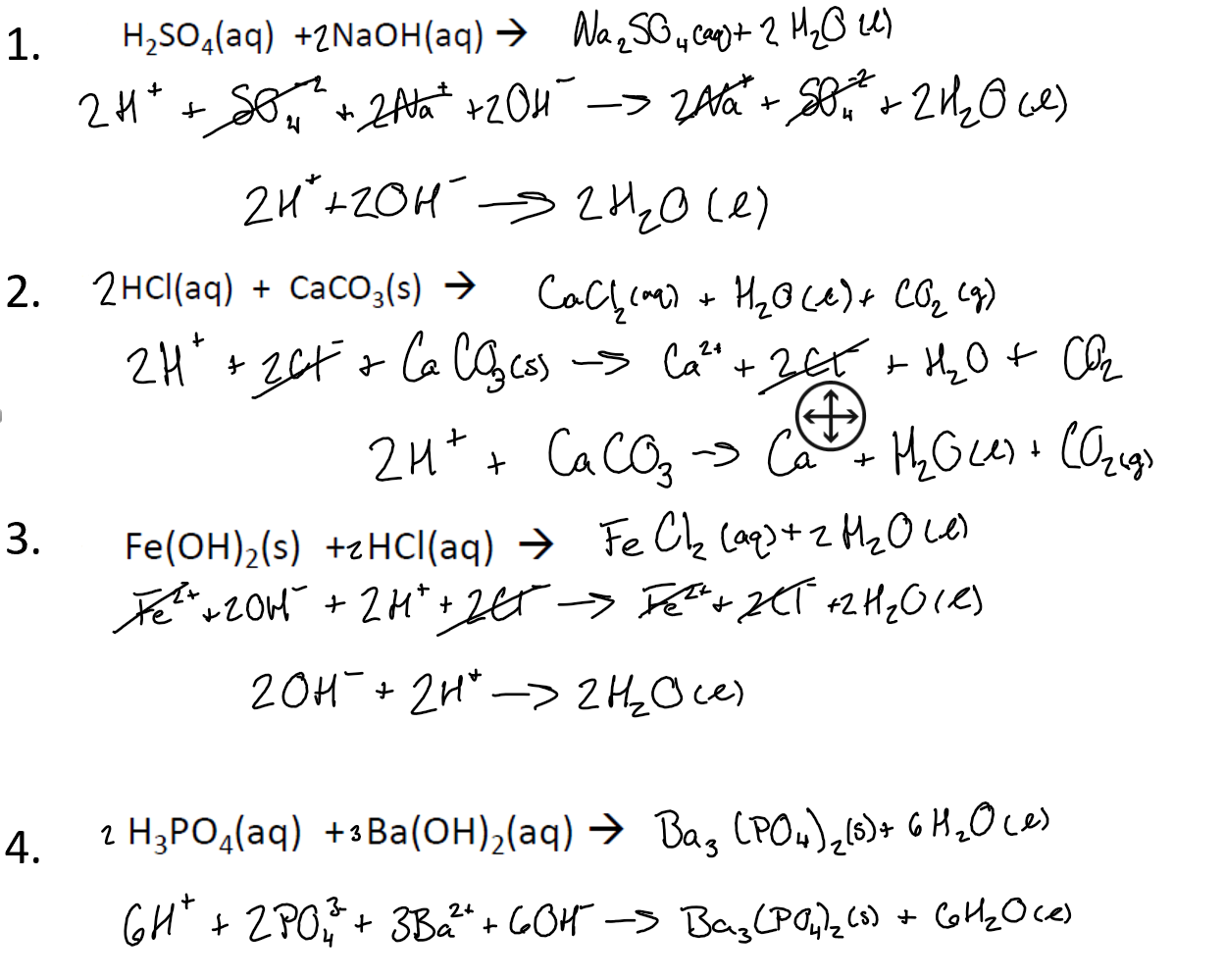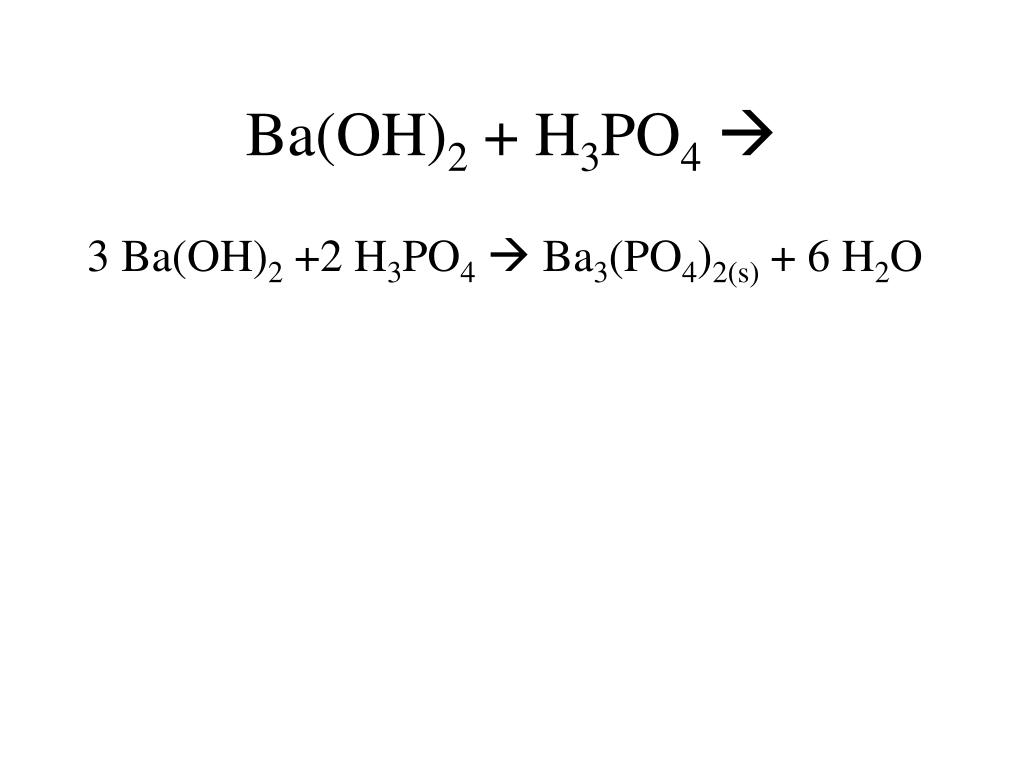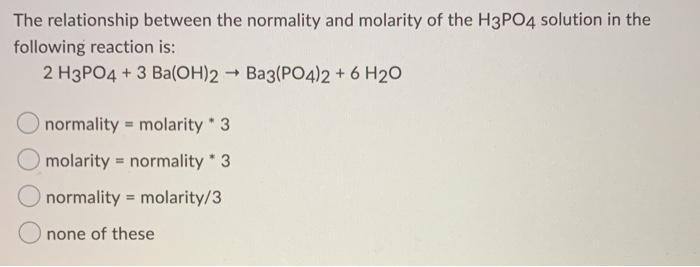SOLVED: Aqueous solutions of barium hydroxide (Ba(OH)2) and phosphoric acid (H3PO4) will react to yield barium phosphate and water. In the balanced equation for this reaction, what is the lowest possible whole
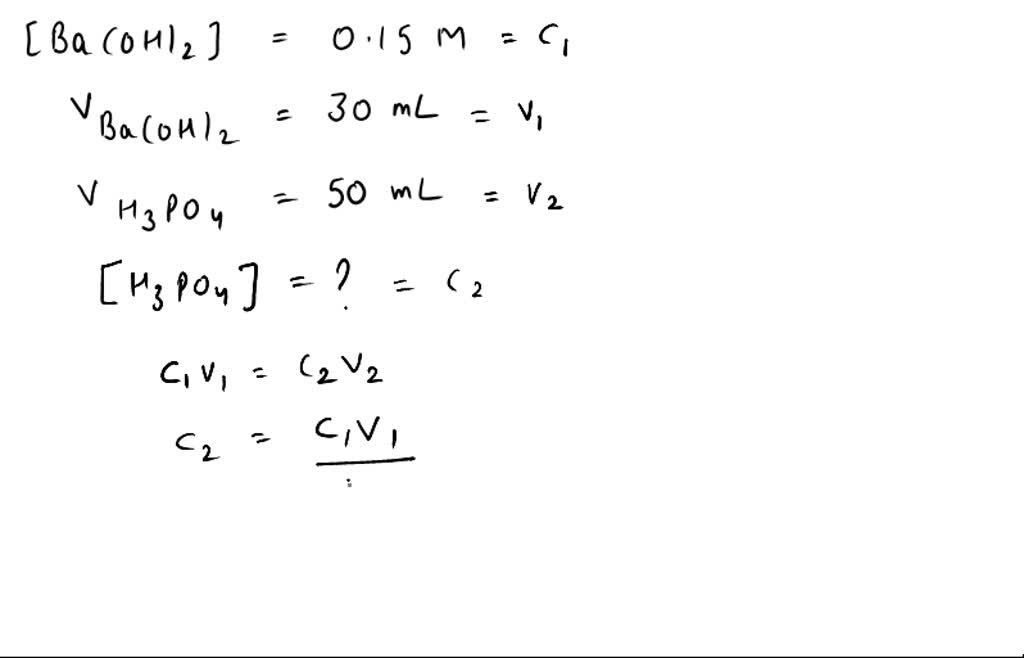
SOLVED: If 30.0 mL of 0.15 M Ba(OH)2 was needed to neutralize 50.0 mL of an H3PO4 solution. What is the concentration of the original H3PO4 solution?

SOLVED: Barium hydroxide and phosphoric acid react as follows: 3 Ba(OH)2(s) + 2 H3PO4(aq) –> Ba3(PO4)2(aq) + 6 H2O(l) If 39.5 g of Ba(OH)2 are allowed to react with 51.0 g of
✓ Solved: What volume of 0.0521 M Ba(OH) 2 is required to neutralize exactly 14.20 mL of 0.141 M H 3...

SOLVED: What would be the total ionic equation for 3Ba(OH)2 (aq) + 2H3PO4 (aq) → Ba3(PO4)2 (s) + 6H2O (aq)? Identify the spectator ions of this reaction.

SOLVED: Question 5 (2 points) In the titration of a 26.07 mL sample of phosphoric acid (H3POA), it requires 37.96 mL of 0.2169 M barium hydroxide (Ba(OH)2) to reach the phenolphthalein endpoint: