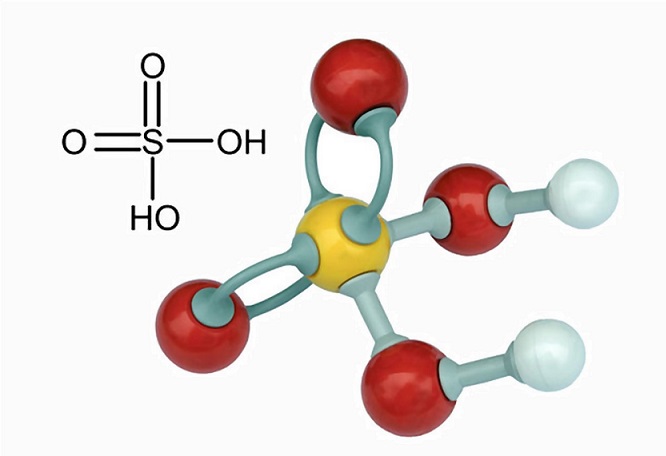
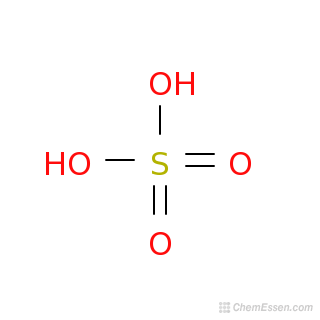
Alumminum hydroxide reacts with sulfuric acid as follows: 2Al(OH)3+H2SO4-->Al2(SO4)+6H2O. Which reagent is the limiting reactant when 0.500 mol Al(OH)3 and 0.500 mol H2SO4 are allowed to react? How ma | Homework.Study.com

SOLVED: In the reaction below; the concentration of H2SO4 was determined from mass of BaSO4: BaCl2(aq) H2SO4 (aq) 5-> BaSOa(s) + 2HCI (aq) A 5.00 mL of diluted sulfuric acid was reacted
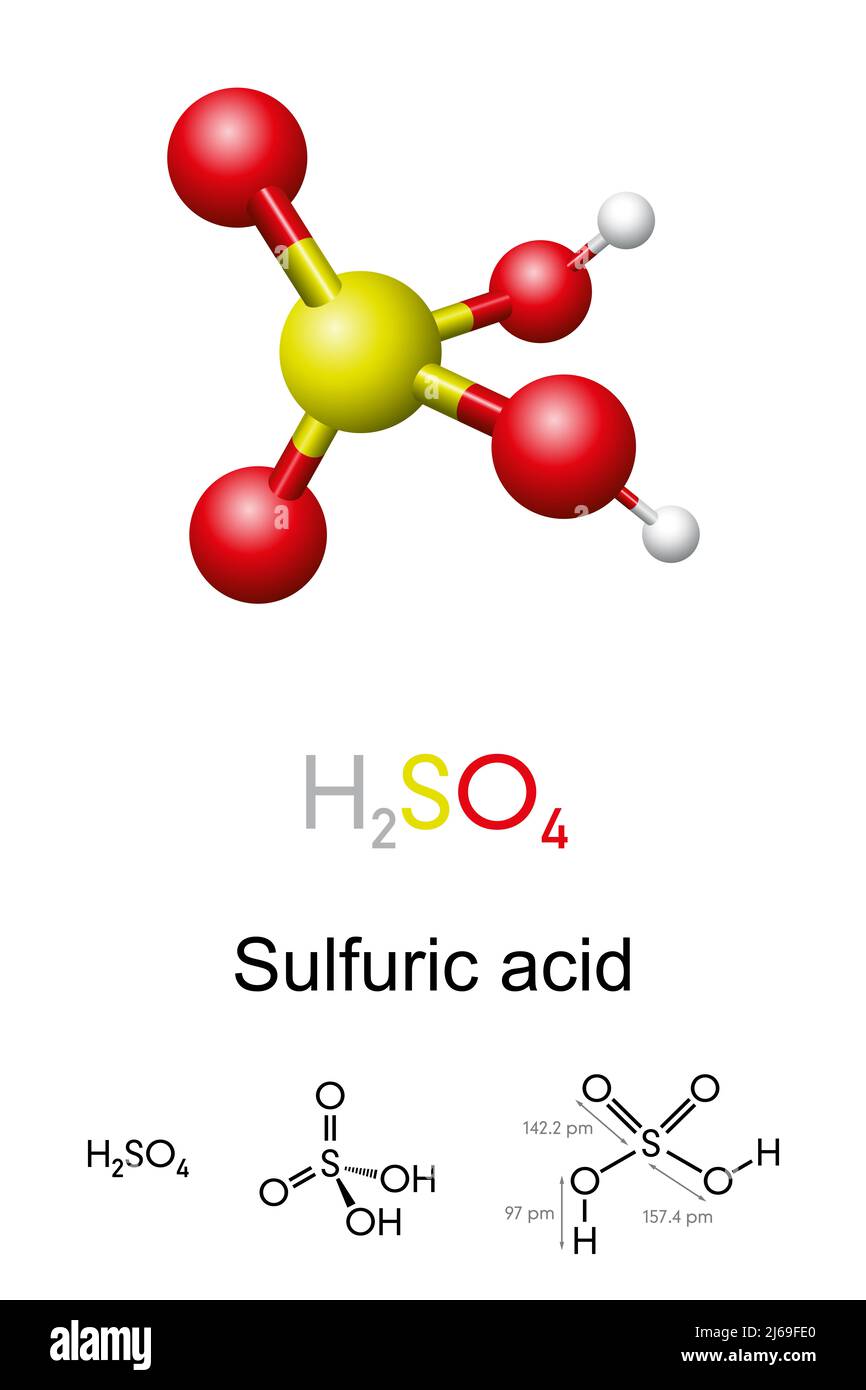
Sulfuric acid, H2SO4, ball-and-stick model, molecular and chemical formula with binding lengths. Known as sulphuric acid, or oil of vitriol Stock Photo - Alamy

What volume of 0.250 mol/L sulfuric acid, H2SO4(aq) is needed to react completely with 37.2 mL of - Brainly.com

Effect of the H2SO4 concentration on the molybdenum recovery. Working... | Download Scientific Diagram

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa



![Sulfuric Acid [H2SO4] Molecular Weight Calculation - Laboratory Notes Sulfuric Acid [H2SO4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/sulfuric-acid-molecular-weight-calculation.jpg)