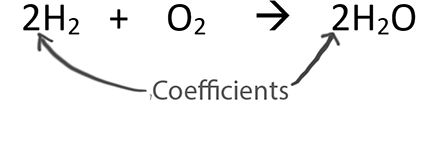Kp for the reaction CO2 + H2 =CO + H2O is found to be 16 at a given temperature. Originally equal number of moles of H2 and CO2 were placed in the
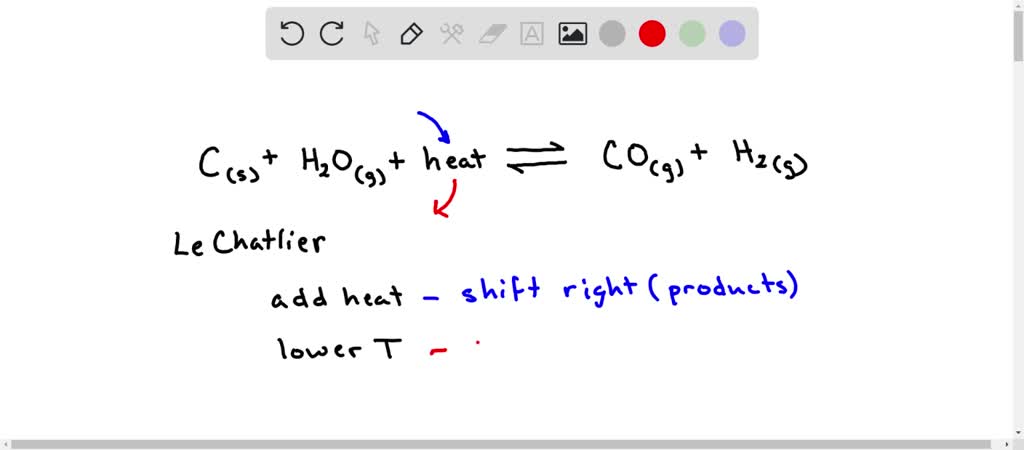
SOLVED: 5. When heated, carbon reacts with water to produce carbon monoxide and hydrogen. C( s ) + H2O(g) + heat ⇌CO(g) + H2(g) What effect does each of the following changes
Free Online Help: Given the following delta H values H2+1/2O2--->H2O delta H =-285.8 H2O2---->H2+O2 delta H = 187.6 Calculate delta H rxn for the following reaction H2O2--->H2O + 1/2O2

n mole each of h2o h2 o2 r tken in closed container at temperature t if y mole of h2 r disasssociated at equillibrium n equillibrium pressure is p the cgt66gee -Chemistry -
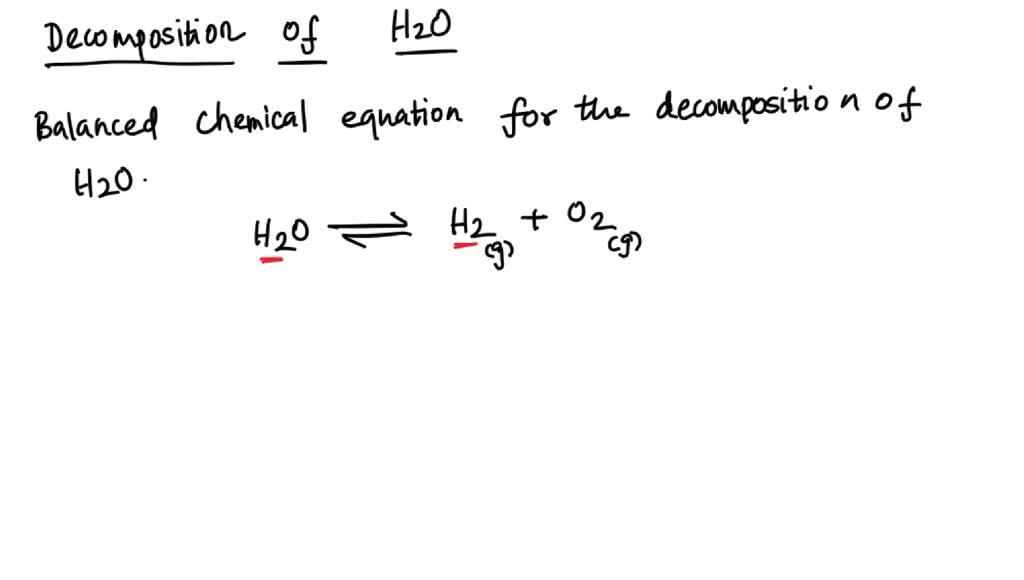
SOLVED: The decomposition of water into hydrogen gas H2 and oxygen gas O2 can be modeled by the balanced chemical equation A) H2 + O2 → H2O B) H2O → H2 +

H2 Evolution from H2O via O–H Oxidative Addition Across a 9,10-Diboraanthracene | Inorganic Chemistry | ChemRxiv | Cambridge Open Engage

n mole each of H2O, H2 and O2 are mixed at a suitable high temperature to attain the equilibrium 2H2O 2H2 + O2 . If y mole of H2O are dissociated and


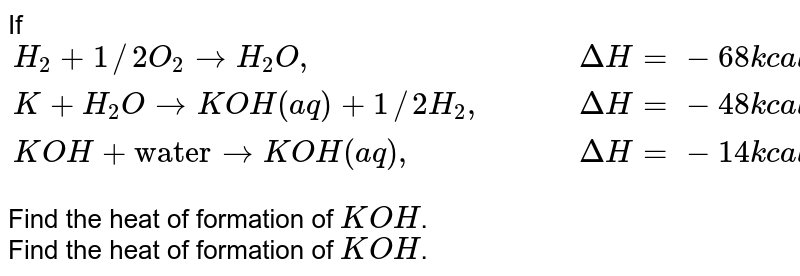
![PDF] Initiation in H2/O2: Rate constants for H2+O2→H+HO2 at high temperature | Semantic Scholar PDF] Initiation in H2/O2: Rate constants for H2+O2→H+HO2 at high temperature | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/59651c1a79d5be73d45e3b492f6f4396965dd05f/5-Table1-1.png)