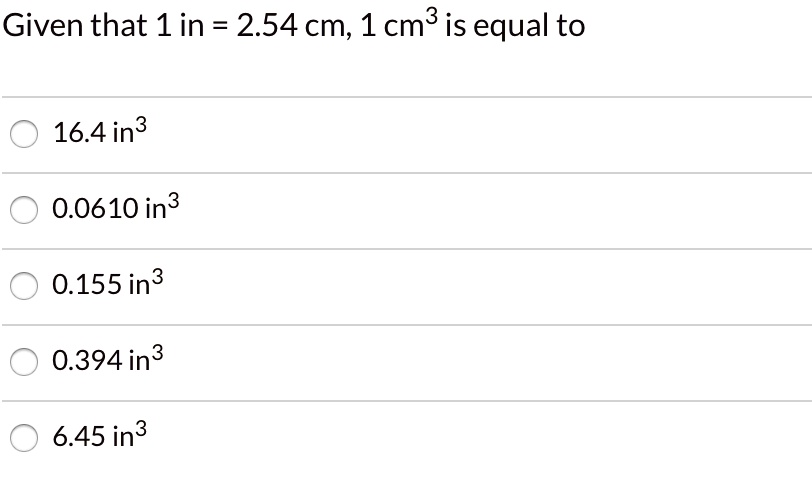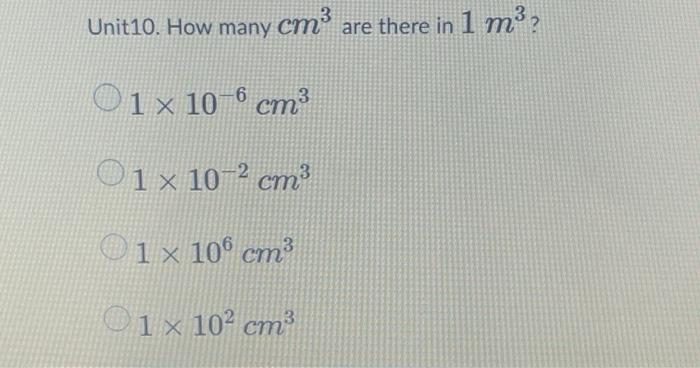
1. 2 Volume – SI derived unit for volume is cubic meter (m 3 ) 1 cm 3 = (1 x m) 3 = 1 x m 3 1 dm 3 = (1 x m) 3 = 1 x m 3 1 L = - ppt download
View question - if we have a value in cm3 nominator and denominator in cm how to get the value in cm2. sound silly .pls help me out

SOLVED: 23. The density of diamond is 3.51 g. cm-3. What is the volume of a diamond with a mass of 0.888 carat? (1 carat = 200 mg; non-SI unit.) 1 0.253 cm3 2) 0.169 cm3 3) 0.0253 cm3 4) 0.0506 cm3
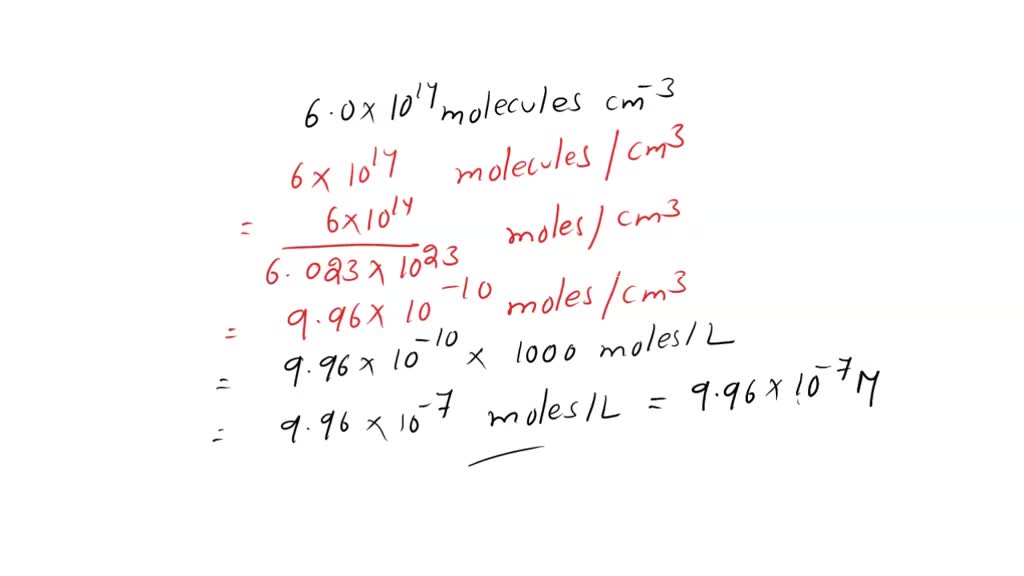
SOLVED: Convert a concentration of 6.0 x 1014 molecules cm-3 to the ppm scale and to the moles per liter(molarity) scale, assuming temperature 25°C and pressure 1 atm.