Introduction to Regulatory Affairs: 3. Roles of the European Directorate for the Quality of Medicines (EDQM) and the European Pharmacopoeia (EP) Commission in the medicines regulations system
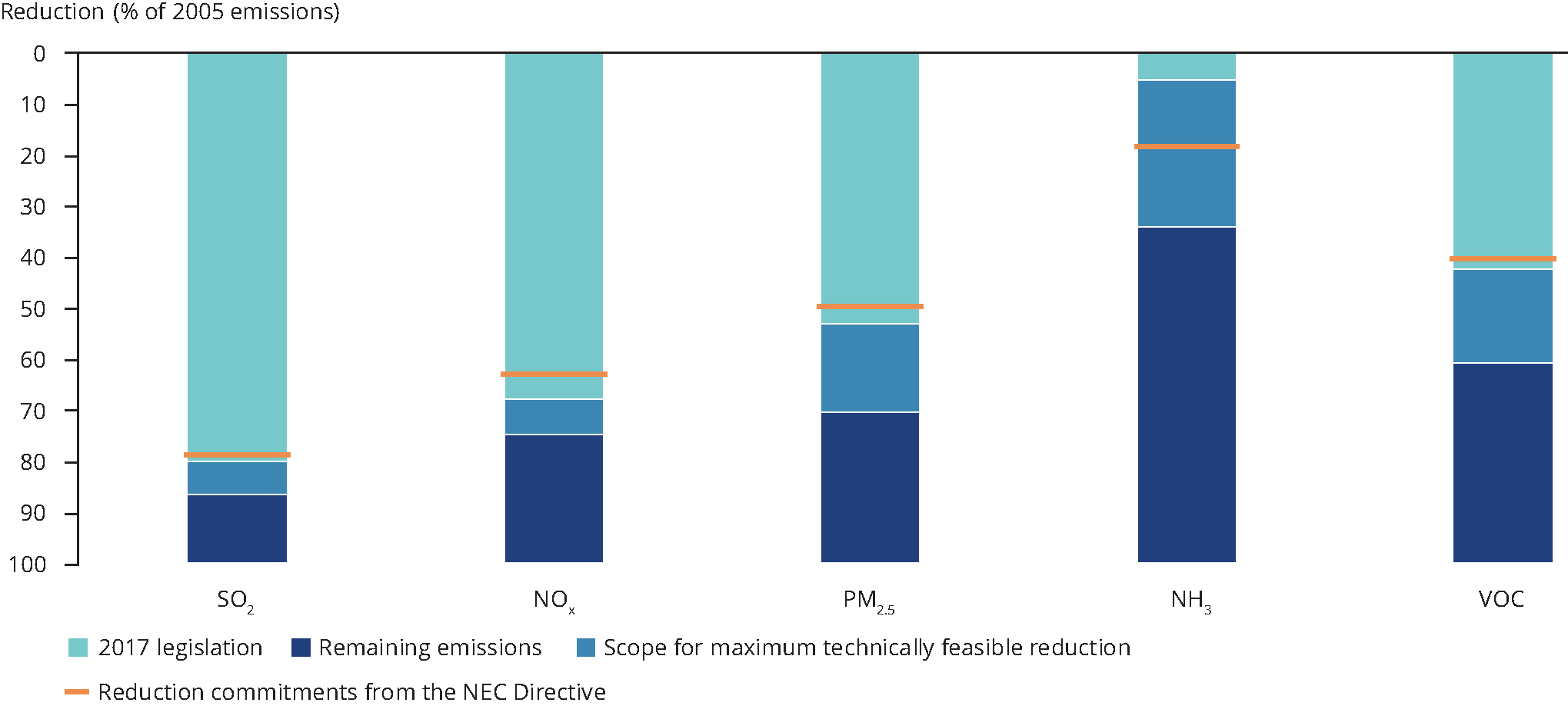
Emission reductions in 2030 relative to 2005 for the REF and CEP scenarios ( EU-28) — European Environment Agency








.jpg)