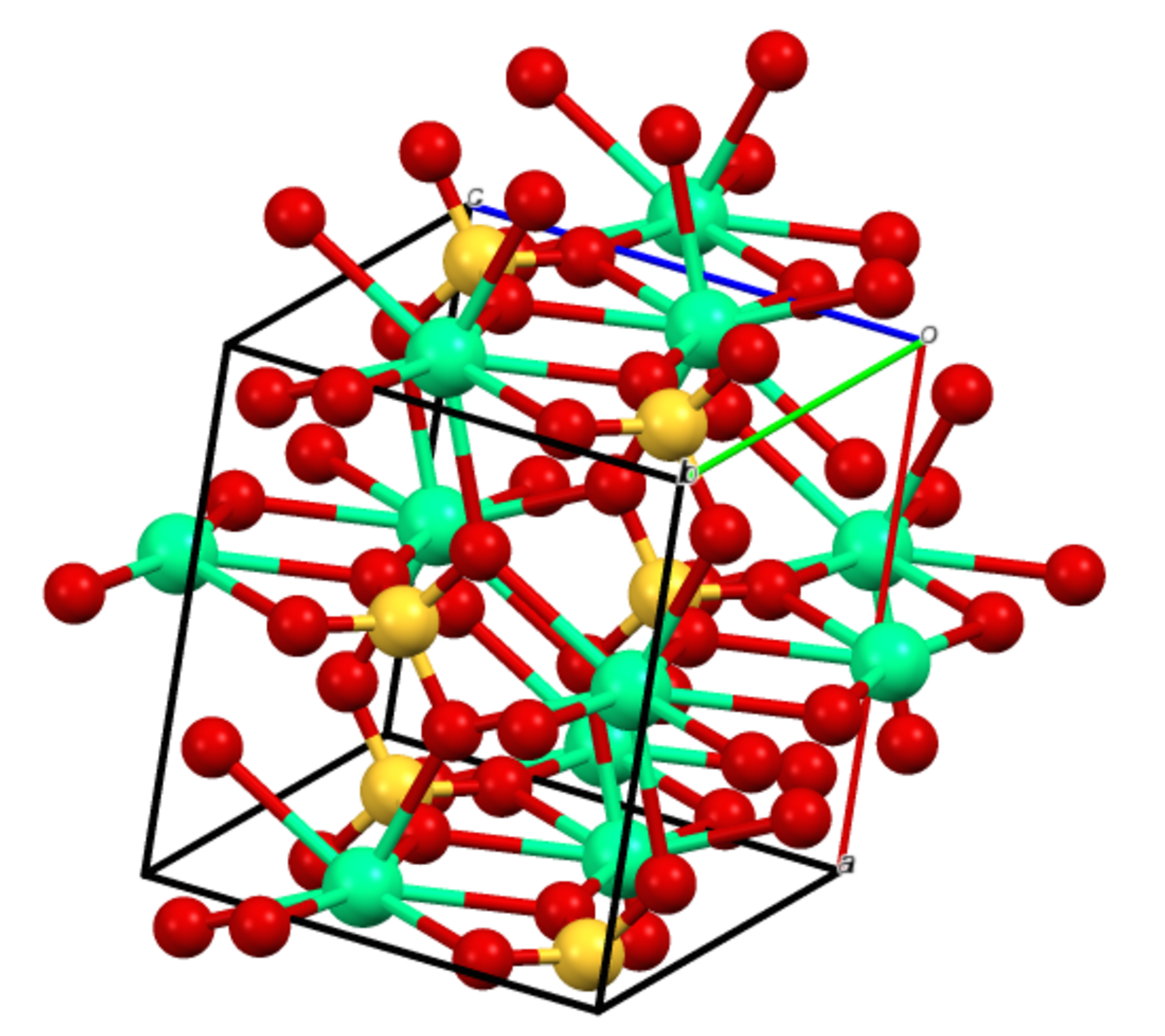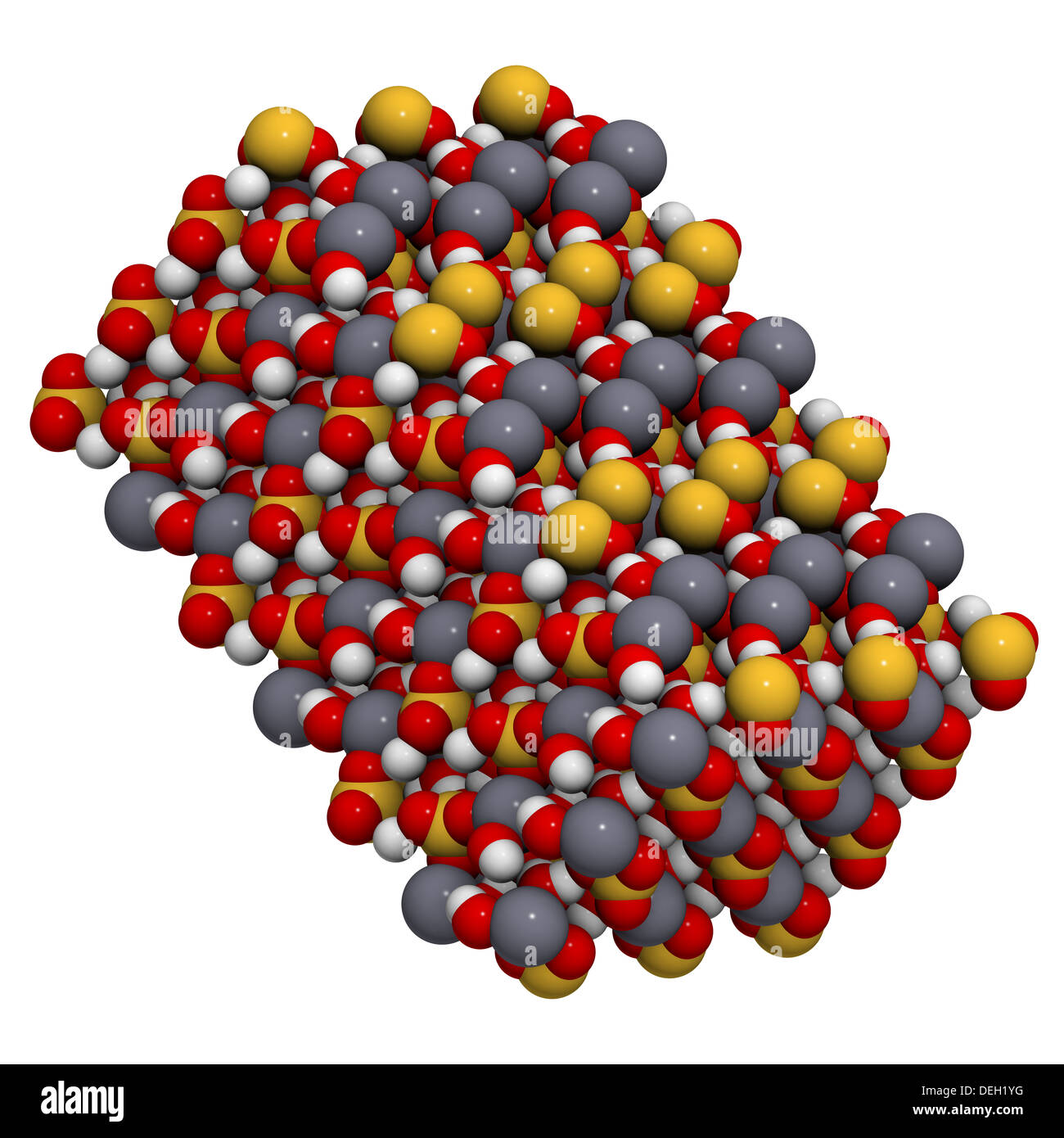
Gypsum (CaSO4.2H2O) mineral, crystal structure. Used for the production of gypsum board, plaster and in fertilizer compositions Stock Photo - Alamy
Influence of structure on the morphology of CaSO4·nH2O (n = 0, 0·5, 2): A molecular simulation study

Thermodynamic Modeling of Calcium Sulfate Hydrates in a CaSO4–H2SO4–H2O System from 273.15 to 473.15 K up to 5 m Sulfuric Acid | Journal of Chemical & Engineering Data

Gypsum (CaSO4.2H2O) Mineral, Crystal Structure. Gypsum Is Used For The Production Of Gypsum Board, Plaster And In Fertilizer Compositions. Stock Photo, Picture And Royalty Free Image. Image 17236747.

Arsenic speciation in synthetic gypsum (CaSO4·2H2O): A synchrotron XAS, single-crystal EPR, and pulsed ENDOR study - ScienceDirect

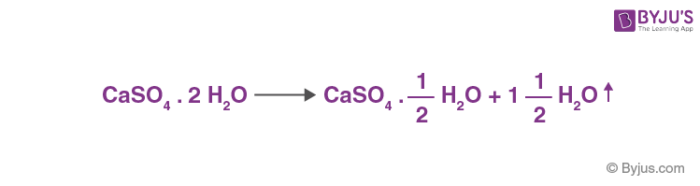
SOLVED: QUESTiON 5 In the following reaction CaSO4*2HzO(s) CaSO4(s) + H2O(g) the anhydrate is CaSOa(s) HzO(g) CaSO4 2H2O(s) none of these
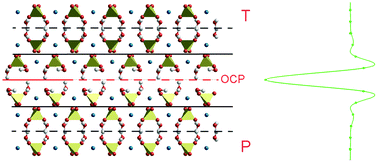
Interfaces structure and stress of gypsum (CaSO4·2H2O) penetration twins - CrystEngComm (RSC Publishing)