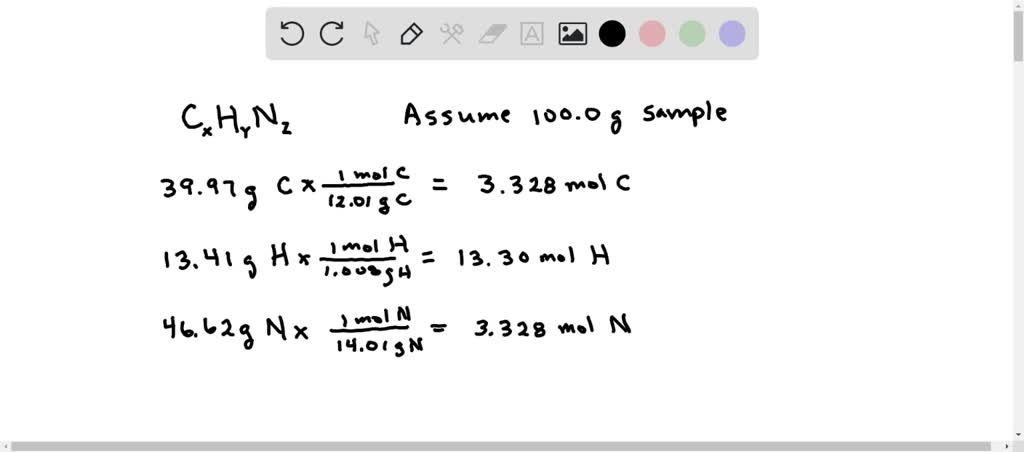
Cu and Ag mole relationship lab - Nathaniel F. Petros CHE 112- Cu and Ag mole relationship lab - Studocu

Large Molecular Weight Nitroxide Biradicals Providing Efficient Dynamic Nuclear Polarization at Temperatures up to 200 K | Journal of the American Chemical Society

Question Video: Calculating the Mass of Solute Needed to Prepare a Solution with a Desired Concentration and Volume | Nagwa

Question Video: Determining the Mass of the Potassium Chloride Analyte When 2g of Silver Chloride Precipitates | Nagwa