![SOLVED: Sa Acetate ion is a better leaving group than methoxide ion in both Swl and Sw2 reaction Explain why: [4 points] CHzO CH; Acetate ion Methoxide ion Sb. Circle the better SOLVED: Sa Acetate ion is a better leaving group than methoxide ion in both Swl and Sw2 reaction Explain why: [4 points] CHzO CH; Acetate ion Methoxide ion Sb. Circle the better](https://cdn.numerade.com/ask_images/75714da48f814e6d9f7e1d00f37c87d1.jpg)
SOLVED: Sa Acetate ion is a better leaving group than methoxide ion in both Swl and Sw2 reaction Explain why: [4 points] CHzO CH; Acetate ion Methoxide ion Sb. Circle the better

Dibutylammonium-acetat -Lösung 0.5 M in H2O, LiChropur™, suitable for ion pair chromatography | Sigma-Aldrich

2,2,2-Trifluoroethyl Acetate as an Electrolyte Solvent for Lithium-Ion Batteries: Effect of Weak Solvation on Electrochemical and Structural Characteristics | The Journal of Physical Chemistry C

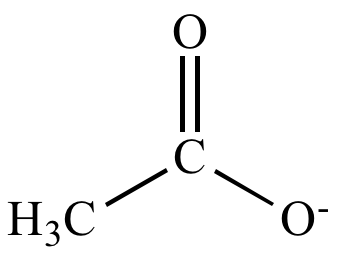
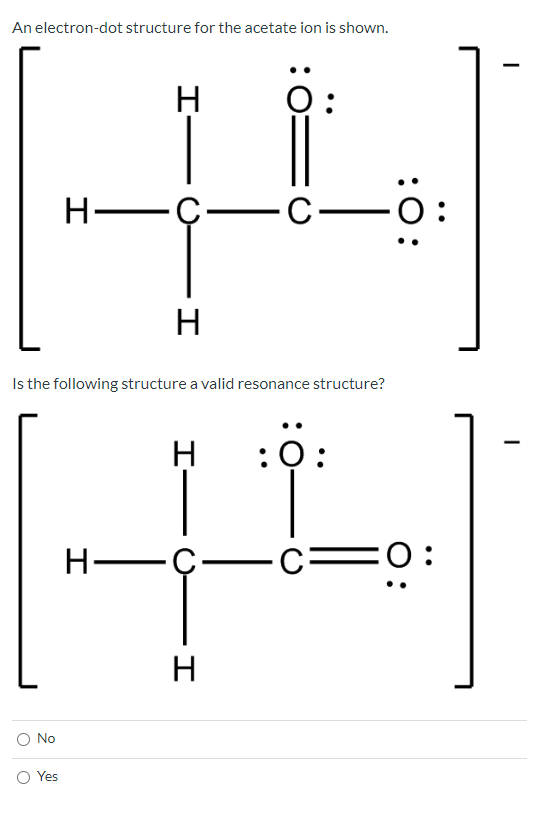
Write the Lewis structure for the Acetate ion, CH_3COO^-. Show all valence electrons and all formal charges. | Homework.Study.com

Simultaneous quantitative analysis of the acetate, formate, chloride, phosphate and sulfate anions in biodiesel by ion chromatography - ScienceDirect
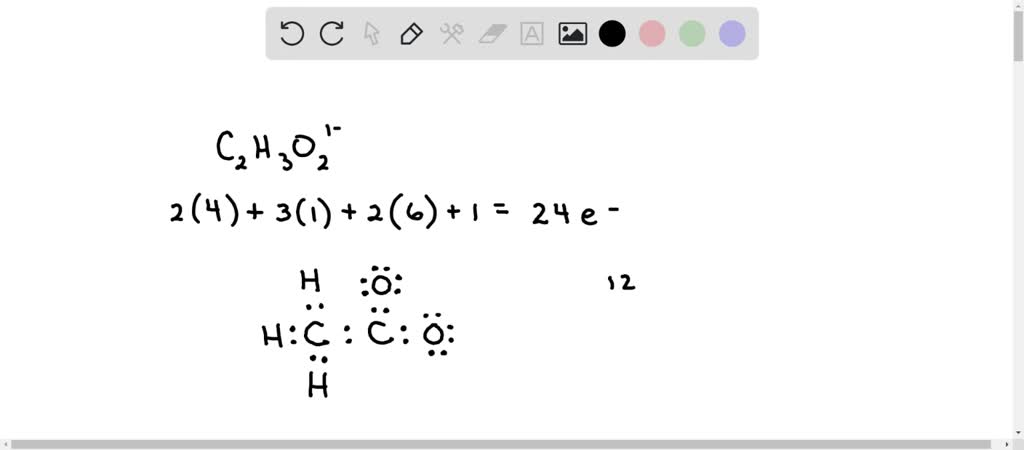
SOLVED: Determine the shape around each central atom in the acetate ion. Draw the Lewis structure first. (enter one of the following: linear, trigonal planar, tetrahedral, trigonal pyramidal, bent, trigonal bipyramidal, seesaw,
![Assertion\\[C{H_3}CO{O^ - }\\] (acetate ion) is more stable than \\[{C_2}{H_5}{O^ - }\\] (ethoxide ion).ReasonElectron delocalisation causes the negative charge in acetate to be shared equally by both oxygens. This type of resonance Assertion\\[C{H_3}CO{O^ - }\\] (acetate ion) is more stable than \\[{C_2}{H_5}{O^ - }\\] (ethoxide ion).ReasonElectron delocalisation causes the negative charge in acetate to be shared equally by both oxygens. This type of resonance](https://www.vedantu.com/question-sets/0962b529-ce35-4eec-b4bf-7221bd3f7a1b1613230999471628212.png)

![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion. Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://cdn1.byjus.com/wp-content/uploads/2019/04/structure-of-acetate.png)